Having battled leukemia twice, Gordon Brooke understands the importance of developing and testing new drugs that will save kids’ lives without causing permanent side effects.
The first time he was diagnosed with acute lymphoblastic leukemia (ALL), Gordon underwent punishing amounts of chemotherapy and a steroid dose so strong it hindered his ability to walk.
“I was bald, ghost pale, skeletal and almost entirely anti-social,” he says.
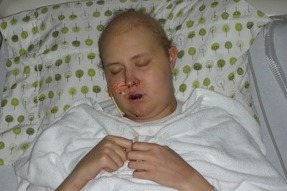
When he relapsed three years later, Gordon’s only hope for survival was a bone marrow transplant (BMT). A BMT is a grueling procedure, in which the patient’s white blood cells are wiped out by high-dose chemotherapy and replaced by a donor’s healthy new blood cells.
The transplant saved Gordon’s life.
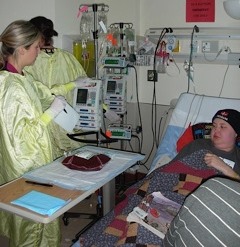
Although BMTs frequently cures cancers and other disorders of blood is the treatment, it is a brutal procedure and the risk of developing complications is high. Roughly 25 to 40 per cent of adult and pediatric BMT patients develop a condition called graft versus host disease (GvHD), which occurs when donor-derived blood cells in a patient see the patient’s body as foreign and triggers an immune response, leading to serious health problems and complications. About 10 per cent of these patients die from GvHD.
Developing new drugs for young people with high-risk cancers is vital and, one day, Carfilzomib may be one of those drugs.
Carfilzomib is currently being evaluated in a phase I clinical trial for difficult-to-treat pediatric cancers at 10 top-tier pediatric hospitals across North America. Open to all eligible children with relapsed leukemia and solid tumours, the study will closely monitor and analyze the side effects of Carfilzomib to determine the maximum dose children can safely tolerate. The trial will cost approximately $3.5 million (U.S.) to administer and is being run through the Pediatric Oncology Experimental Therapeutics International Consortium (POETIC).

Previously tested and approved for use in adults, Carfilzomib is already showing promise for treating adult patients with multiple myeloma — a high-risk leukemia. Studies on Carfilzomib in a University of Calgary lab revealed a similar penchant for killing pediatric cancer cells. These findings from Dr. Narendran’s lab were presented last year at the American Association of Cancer Research.
Dr. Narendran and Dr. Jessica Boklan of Phoenix Children’s Hospital are the principal investigators in the clinical trial and Dr. Tony Truong, co-principal investigator at the Alberta Children’s Hospital, and U of C clinical research staff Karen Mazil and Pina Giuliano, are helping monitor side effects and analyze findings from the study.
Carfilzomib is a proteasome inhibitor, meaning it stops the proteasome mechanism in cancer cells that helps cancer to survive. Because cancer cells divide and multiply so quickly they make a lot of mistakes, leading to abnormally formed cells. These poorly formed cells put stress on the cancer and threaten its survival. To get around this, the cancer cell uses proteasomes to repair its mistakes. By inhibiting the proteasome, Carfilzomib helps the cancer kill itself by its own imperfections.
“The beauty of this drug is it’s not cancer-specific,” says Dr. Narendran a pediatric cancer researcher with the Experimental and Applied Therapeutics (ExpAT) program at the Childhood Cancer Research Program at the Alberta Children’s Hospital Research Institute and the University of Calgary. “It blocks a mechanism that is vital to pretty much all malignant cells, so it can be used against different cancers. Normal cells don’t make so many mistakes, so this drug will be less toxic to normal cells, which may mean fewer side effects for children.”
Another promising attribute of Carfilzomib is that it works synergistically with other chemotherapies to boost their ability to kill cancer cells. This is especially important for chemo-resistant cancers.
“It’s kind of a safety net,” says Dr. Narendran. “Because the chemo agents and their toxicity are already known to us, we can use these chemotherapies with Carfilzomib to boost their potency for better outcomes.”
“What we know so far comes only from adult patients or from laboratory studies against pediatric cancer cells,” says Dr. Narendran. “We do not know if it will actually offer any benefit to pediatric cancer patients yet.”
As a phase I clinical trial, the study is not intended to cure children, but to understand its toxicities and determine suitable doses for treating children in the future.
Still, it is the first important step in a long quest to find safe and effective treatments for children with incurable cancers. If it lives up to its potential, Carfilzomib may one day offer hope for children with high-risk cancers.

Fortunately, Gordon Brooke has not experienced complications from the BMT he received. He’s healthy and strong today and with the help of a Kids Cancer Care scholarship Gordon is studying pharmacology at the University of Alberta. His experiences with cancer and chemotherapy inspired him to pursue a career in pharmacological research. One day, Gordon hopes to use his expertise to further refine chemotherapies to help young people like him.
















